The CIMScan/SmartScan web application does currently rely on Adobe Flash as part of its function. Flash will reach end of life at the end of 2020.
Learn MoreSmartScan Software
SmartScan for Temperature and Humidity Monitoring in Healthcare Facilities
Hospital environments require secure, 24/7 monitoring to maintain drugs, vaccines, blood and tissues, and critical medical devices within specified
Learn MoreYour Continuous Monitoring Questions Answered
Your Continuous Monitoring Questions Answered When choosing a Continuous Monitoring System (CMS) it’s good to start asking questions to be sure
Learn MoreSmartScan is the Simple Solution for Vaccine Storage
“A refrigerator is ONLY as good as the temperature monitoring system inside” – CDC Depending on your facility and application vaccines can be
Learn MoreYour 24/7 Web Based Monitoring System Solution
Imagine vaccines stocked for flu season, embryos frozen for fertility treatments, new pharmaceuticals being formulated… how do you insure that your
Learn MoreChoosing your Continuous Monitoring System
It is important to weigh your options when choosing the right continuous monitoring system and we have a list of important questions for you to keep
Learn MoreBenefits of Continuously Monitoring Your Critical Assets
Have you been considering researching and investing in a Continuous Monitoring System like SmartScan? Let us help you hone in on the benefits and
Learn MoreSmartScan Generates Custom Reports with Ease
SmartScan can automatically generate a comprehensive batch report containing multiple processes for a pharmaceutical or medical device manufacturer.
Learn MoreMust be able to Interface with Other Systems
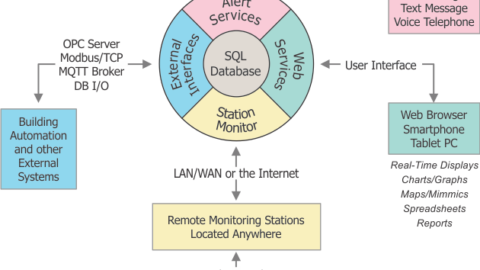
Measurement data, including values, alarm status, and a timestamp can be easily exported to other systems using OPC (SmartScan acting as the server),
Learn MoreMust Support the Use of 3rd Party Instruments
Many third party instruments provide one or more analog outputs for their measurements. These can be easily tied to a SmartScan remote device
Learn MoreMust be a fully validated system
The SmartScan Validation Templates consist of four documents in Microsoft Word or PDF format. The documents have been designed to minimize the effort
Learn MoreWhat Is Gmp Compliance And Do I Need It?
Good manufacturing practices (GMP) are the practices required in order to conform to guidelines recommended by agencies that control authorization and
Learn MoreHighly Scalable : Your System Grows and Changes with You
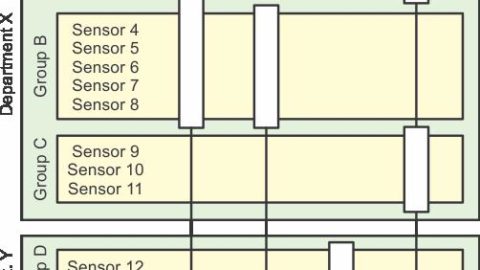
SmartScan is highly scalable from just a few monitoring points to thousands. Each numeric monitoring point can have a measurement value
Learn MoreMust Be Standards Based
SmartScan server software has been created using GAMP 5 guidelines using standard programming languages and methodologies to run under standard
Learn MoreMust Meet Regulatory Requirements
SmartScan meets or exceeds the guidance published in FDA 21 CFR Part 11 which covers the security (preventing change) of data stored in electronic
Learn More