One System Continuously Monitors the Environment in Cleanrooms as well as the Condition of the Supporting Infrastructure
SmartScan can be used to implement a Clean Room Monitoring System that is fully cGMP-Compliant including Temperature and Humidity Monitoring, Differential Pressure Monitoring, and Airborne Particle Monitoring. The system meets ISO 14644 requirements and conforms to the data integrity stipulations specified in FDA 21 CFR Part 11. SmartScan is a Facility Monitoring System that …
- Can be hosted on your server or ours in the cloud and can be accessed from anywhere using a simple web browser or smart phone
- Meets or exceeds the monitoring requirements specified by the FDA, EU, and other government regulators.
- Automatically maintains records for auditors
- Detects abnormal conditions and sends alerts via email, text, pager or voice telephone
- Contains powerful analysis and reporting tools along with user-customizable reports
- Built around a standard SQL database and is scalable from a few monitoring points to thousands
- Fully user-configurable/maintainable
- Contains the capabilities and data acquisition components necessary to monitor ultrapure water systems or do sophisticated Process Monitoring.
- A complete line of instrumentation is available or interface with 3rd party devices via analog signals or using the Modbus protocol
CONTINUOUSLY MONITOR
- Temperature
- Temp/Humidity
- Differential Pressure
- Airborne Particles
- Air Flow
- Door Open/Ajar
- Refrigerator Temperature
- Freezer Temperature
- Ultra-Pure Water Systems
- Vacuum System
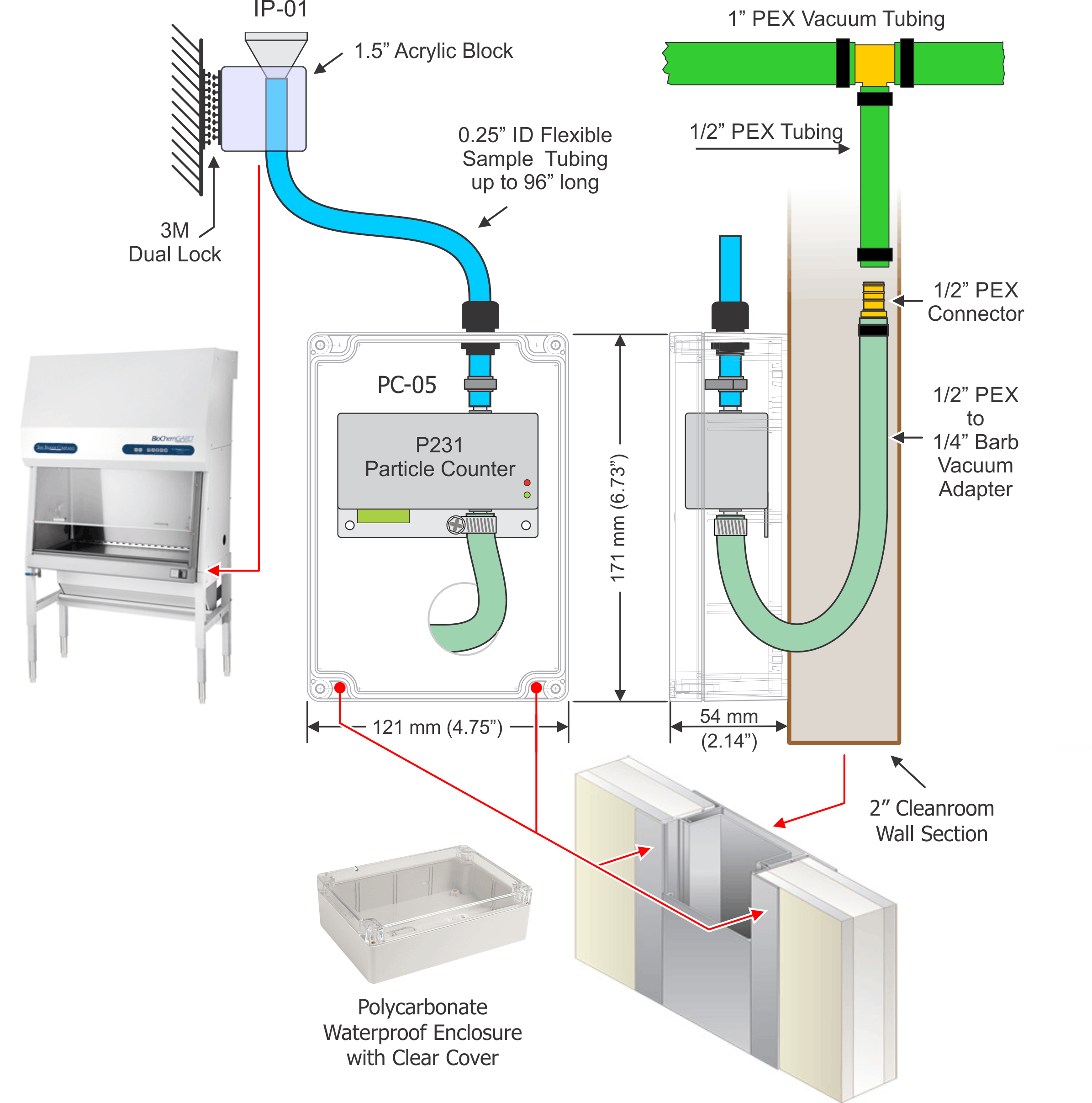
TYPICAL CLEANROOM INSTRUMENTATION

CEILING-MOUNTED SENSORS

PARTICLE COUNTERS
Lauren Lawson says
Hello, I am interested in a price quote of your smartscan product. Also how long would it take for instillation of our sterile compounding pharmacy?
Leigh Basso says
Good morning Lauren if you can provide your email address I would be happy to get you some information . I may need some more details to give you an accurate estimation for installation but would love to get a solution together for you. Thanks so much
Leigh Basso
Leigh.basso@smartscanmonitoring.com